
Physicochemical Properties, Mineral and Free Amino Acid Contents, Antioxidant Activities, and Sensory Quality of Makgeolli Containing Jerusalem Artichoke Water Extracts
Abstract
This study examined the effects of Jerusalem artichoke (JA) hot-water extract (JAE) on the quality characteristics of Makgeolli. The purpose was to reduce undesirable discoloration, including browning, that can occur when raw JA is added directly, while enriching Makgeolli with JA-derived soluble nutrients and bioactive compounds. Accordingly, JAE was prepared using the hot-water extract of fresh JA at 0, 5, 10, 15, and 20% (w/w) based on the rice weight. The entire volume of JAE obtained at each level was then added to the Makgeolli mash. The physicochemical properties (color, mineral, free amino acid, and total polyphenol contents), antioxidant activities, and sensory quality were evaluated. The ethanol content (6.08%—6.12%), pH (4.02—4.07), and titratable acidity (0.18%—0.24%) were similar across treatments. By contrast, the soluble solids (°Brix) increased slightly from 3.1% in the control to 3.4%—4.2% in the JAE-added samples. In proportion to the addition of JAE, the L* values of the Hunter color decreased from 52.37 to 40.00; the a* values increased from —0.32 to 3.21, and the b* values showed the largest rise. The K and Ca levels were 120.11—145.11 mg/L, Zn decreased from 80.29 to 32.00—35.18 mg/L, while the Fe and Mg levels increased, reaching 89.71 mg/L and 79.77 mg/L at 20% JAE. The Mn and Cu contents remained stable, and no toxic heavy metals (As, Hg, Cd, and Pb) were detected. The essential amino acids levels followed the order, Phe > Lys > Trp > Val > Ile > Met > Thr > Leu > His, and the key free amino acids (Ala, Glu, and Arg) and functional amino acids, such as L-ornithine, γ-aminobutyric acid, increased in proportion with JAE addition. The total polyphenol content increased from 312.11 mg% to 326.23—404.31 mg%, enhancing the DPPH and hydroxyl radical scavenging activities. The sensory evaluation indicated significantly higher scores in taste, color, aroma, and overall acceptability for Makgeolli with 5%—20% JAE compared to the control. Thus, JAE addition can simultaneously enhance the nutritional, functional, and sensory qualities of Makgeolli, supporting its potential as a functional fermented beverage. The optimal percentage JAE addition, as determined by sensory scoring, was approximately 15%.
Keywords:
Makgeolli, Jerusalem artichoke water extract, fermentation, free amino acids, antioxidant activity, sensory qualityINTRODUCTION
Jerusalem artichoke (Helianthus tuberosus L.) is a perennial plant belonging to the Asteraceae family. In Korea, it is commonly known as ttungttanji or dwaeji-gamja, and it is distributed widely because of its high adaptability to various climatic conditions (Kim HS & Hwang ES 2022a). Jerusalem artichoke contains a high inulin content (approximately 70%—75% on a dry matter basis), while its fresh form consists of approximately 80% moisture with an inulin content of 14%—15% (Nair KK et al 2010; Shin SH et al 2012). Inulin is a type of dietary fiber that is insoluble in alcohol but slightly soluble in water. Although extensive studies have been conducted on its use as a bioethanol production substrate (Lim SH et al 2010; Liu ZX et al 2015; Wang YZ et al 2015; Khatun MM et al 2017), it has recently attracted attention as an antidiabetic material because of its blood glucose-lowering effects even under high-fat diet conditions (Shao T et al 2020).
In addition to the glycemic control effects of inulin, Jerusalem artichoke contains organic acids, minerals, pigments, amino acids, and polyphenolic compounds, which have been reported to exhibit immunomodulatory, cardiovascular health-promoting, and antioxidant activities (Lee EH et al 2007; Sawicka B et al 2020; Shao T et al 2020; Li J et al 2022). Accordingly, several studies have been conducted to utilize the functional properties of Jerusalem artichoke in various food products, including fermented milk (Guo X et al 2018), jelly (Kim HS & Hwang ES 2022a), Sulgidduk (Park HS 2010; Shin MH & Chung NY 2019), and brown rice porridge (Kim HS & Hwang ES 2022b).
Alcoholic fermentation of Jerusalem artichoke differs from conventional starch-based fermentation, in which saccharification is induced by α- or β-amylase from nuruk. Instead, the substrate in Jerusalem artichoke is inulin, which can be saccharified by the inulinase produced by specific yeast strains. The saccharified inulin is then fermented by yeast, similar to monosaccharide fermentation (Guiraud JP et al 1983; Sheng J et al 2007; Chi Z et al 2009; Choi JS et al 2022; Choi JS et al 2023). Various forms of Jerusalem artichoke can be considered for Makgeolli production, including raw, heat-treated, or extracted forms. Choi JS et al (2023) reported that heat-treated Jerusalem artichoke resulted in favorable fermentation efficiency and product quality. Preliminary experiments conducted in this study showed that using raw Jerusalem artichoke or its dried powder together with rice during steaming resulted in considerable discoloration during fermentation or during product storage, reducing the marketability. Therefore, this study employed a water extract of Jerusalem artichoke, given that major functional components, such as inulin, are water-soluble, aiming to prevent discoloration in the final fermented product, while also inducing color stabilization through heat extraction. In addition, to avoid the dilution of flavor, color, and components that typically occurs due to post-fermentation dilution for alcohol adjustment in conventional Makgeolli, fermentation was conducted with a higher dilution ratio. Accordingly, the physicochemical properties (alcohol content, pH, titratable acidity, soluble solids (°Brix), and color), mineral composition, free amino acids, total polyphenol content, antioxidant activity, and sensory quality of the final fermented Makgeolli were investigated and compared according to the levels of Jerusalem artichoke water extract added.
MATERIALS AND METHODS
1. Materials
Polished white rice (91% polished rice, Seonsan rice, Hana Agricultural Cooperation, Korea) was used as the raw material. Nuruk with a saccharifying power of ≥300 (Jinju Nuruk, Jinju, Kyungnam, Korea) was used as the fermentation starter. Jerusalem artichoke (Helianthus tuberosus L.) was purchased from a local farm in Gumi, Korea. Brewing yeast (Saccharomyces cerevisiae KMBL 7001; KACC 93363P) was used, and commercial bottled water (Samdasoo, Jeju Special Self-Governing Province Development Co., Korea) was used as brewing water.
2. Preparation of Jerusalem Artichoke Water Extract and Makgeolli Fermentation
The Jerusalem artichoke water extract (JAE) was prepared by thoroughly washing the tubers, slicing them to approximately 5 mm thickness, and extracting with five times their weight of water using a pressure cooker at 12 psi for 30 min. The extract was filtered through gauze. The first fermentation stage for Makgeolli was divided into treatments with no JAE addition (0%) and with JAE added at levels of 5%, 10%, 15%, and 20% based on the rice weight. Fig. 1 presents the brewing and fermentation process of Makgeolli supplemented with JAE. For the Mitsul (primary brewing phase), 10 kg of rice was soaked in tap water at 20℃ for 8 hr, draining for 2 hr, and steaming for 1 hr. After cooling to 20℃, the rice was thoroughly mixed with Nuruk. Ground Nuruk was added at 15% (w/w) of rice weight (1.5 kg for each brewing phase) and mixed thoroughly with steamed rice in a 50 L stainless steel container. Yeast was added at 0.5% (w/w) of the rice weight (50 g during the first fermentation), dissolved in a small amount of brewing water, and the total water volume was adjusted to 16 L. Fermentation was conducted at 25℃. After two days of the Mitsul, Deotsul (secondary brewing phase) was initiated by adding all ingredients except yeast, followed by fermentation for six additional days under the same conditions. The fermented Makgeolli was filtered through cotton cloth and used for physicochemical, color, antioxidant, and sensory analyses. The samples for mineral and amino acid analyses were pasteurized at 85℃ for 5 min and stored at 4℃.
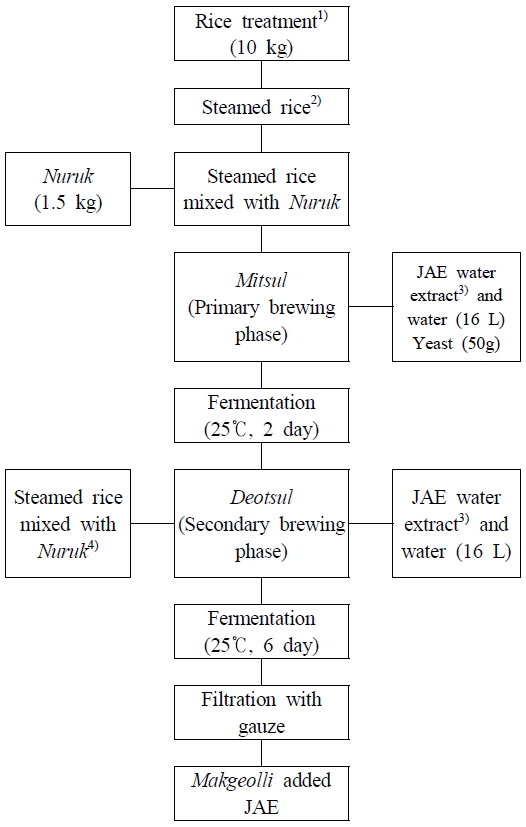
Brewing and fermentation process of Makgeolli supplemented with JAE.1,2) Rice was thoroughly washed, soaked in water for 8 h, and steamed under high pressure. The steamed rice was allowed to cool at room temperature and then mixed with powdered Nuruk. 3) JAE (Jerusalem artichoke hot-water extract) was prepared by adding five volumes of water, boiling at 12 psi for 30 min, and filtering through gauze. JAE was added at 0, 5, 10, 15, and 20%, and the amount of rice was reduced according to the calculated moisture contribution from the added JAE. The total liquid volume (water + JAE) was adjusted to 16 L. The rice amounts were 9.83 kg (5%), 9.66 kg (10%), 9.49 kg (15%), and 9.32 kg (20%). 4) For the secondary brewing phase, steamed rice with Nuruk was prepared using the same washing-soaking-steaming procedure as in steps 1 and 2, allowed to cool at room temperature, and then mixed with powdered Nuruk.
3. Determination of the Alcohol Content, pH, Titratable Acidity, Soluble Solids (°Brix), and Color
The alcohol content was determined by mixing 50 mL of filtered Makgeolli sample with 50 mL of deionized water, followed by distillation. The distillate was collected in a 100 mL graduated cylinder, adjusted to 15℃, and measured using an alcohol hydrometer (AHS-01, Hwngyeong Tech Co., Ltd, Seoul, Korea). The pH was measured using a pH meter (Model 250; Beckman Coulter Inc., Fullerton, CA, USA). The titratable acidity was determined by diluting 5 mL of filtered Makgeolli sample (Whatman No. 2) with 20 mL of deionized water, followed by titration with 0.1 N NaOH (f = 1.000) on a vortex mixer equipped with a pH meter until pH 8.2 was reached. The amount of NaOH consumed was converted to the lactic acid content and expressed as % (w/v). The soluble solids (°Brix) were measured using a digital refractometer (PR-32a, Atago Co., Ltd, Japan). The color values were measured using a chroma meter (CR-300, Minolta Corp., Osaka, Japan) and expressed as the Hunter L* (lightness), a* (redness/greenness), and b* (yellowness/blueness) values. A Minolta calibration plate (YCIE=94.5, XCIE=0.3160, YCIE=0.330) and a HunterLab standard plate (L*=97.51, a*=—0.18, b*=+1.67) were used to standardize the instrument using a D65 illuminant.
4. Mineral Content, Free Amino Acids, Total Polyphenols, and Antioxidant Activities
The mineral content was analyzed using a modification of the method reported by Skujins S (1998) and Vyacheslavov AV et al (2018). Briefly, 0.5 mL of the sample was placed in a 100 mL beaker, mixed with 15 mL of 65% HNO3 (supra pure grade), and heated until complete digestion to a clear solution. The digested solution was diluted with an equal volume of distilled water and analyzed using an inductively coupled plasma atomic emission spectrometer (ICP-AES; Varian Vista, Varian Australia, Victoria, Australia). The operating conditions were as follows: RF power 1.2—1.3 kW, plasma gas flow rate 15 L/min, auxiliary gas flow rate 0.2 L/min, and nebulizer gas flow rate 0.8 L/min. Emission lines (nm) were: Na 589.592, K 766.490, Ca 317.933, Zn 213.856, Fe 259.940, Mg 279.079, Mn 259.373, Cu 324.754, As 189.042, Hg 194.163, Cd 228.802, and Pb 220.353. Calibration curves showed R2=0.9990. Mineral analysis was performed using the Makgeolli filtrate obtained after fermentation was completed and the sample was filtered through cotton cloth. Therefore, the mineral data reported in this study represent the concentrations of minerals retained in the liquid fraction after filtration. The free amino acids were analyzed using the method described by Shin SH et al (2012). Filtered Makgeolli (50 mL; Whatman No. 2) was concentrated using a rotary evaporator (R-100, Büchi Labortechnik, Switzerland), dissolved in 10 mL of 0.12 N lithium citrate buffer (pH 2.2), and mixed with 0.2 g of sulfosalicylic acid. The mixture was stored at 4℃ for 1 hr and filtered through a 0.2 μm membrane filter (BioMed, USA). The filtrate (1 mL) was diluted with an equal volume of lithium citrate buffer and analyzed using an amino acid autoanalyzer (Hitachi L-8900, Tokyo, Japan). Identification and quantification were performed using standard free amino acid and derivative kits (Waters Co., USA) based on the peak areas. The total polyphenol content was determined using the method reported by Singleton VL et al (1999). Makgeolli samples were filtered through cotton cloth and then through Whatman No. 3 filter paper. A 100 μL aliquot was mixed with 2 mL of 2% sodium carbonate and 100 μL of 50% Folin-Ciocalteu reagent (Sigma-Aldrich Corporation, St. Louis, MO, USA) on a vortex mixer. The absorbance was measured at 720 nm using a UV-Vis spectrophotometer (Hitachi U-3900, Hitachi Ltd, Tokyo, Japan). The total polyphenol content was calculated using a gallic acid calibration curve and expressed as mg% (w/v). The electron-donating ability was measured using the method reported by Blois MS (1958). A 200 μL aliquot of sample solution used for polyphenol analysis was mixed with 800 μL of 0.4 mM DPPH (1,1-diphenyl-2-picrylhydrazyl) solution, allowed to react for 10 min, and the absorbance was measured at 525 nm. In the control, the sample was replaced with an equal volume of distilled water. The electron-donating ability (%) was calculated as follows:
The hydroxyl radical scavenging activity was measured using the method reported by Chung SK et al (1997). A reaction mixture containing FeSO₄·7H₂O with 10 mM EDTA and 10 mM 2-deoxyribose was mixed with 200 μL of Makgeolli sample, and the total volume was adjusted to 1.8 mL with 0.1 M phosphate buffer (pH 7.4). After adding 200 μL of 10 mM hydrogen peroxide, the mixture was incubated at 37℃ for 4h. Subsequently, 1 mL of 2.8% trichloroacetic acid (TCA) and 1 mL of 1% thiobarbituric acid (TBA) were added, and the mixture was heated in a boiling water bath for 10 min. After cooling to room temperature, the absorbance was measured at 532 nm, and the scavenging activity (%) was calculated.
5. Sensory Evaluation
A sensory evaluation was conducted using a panel of 20 students (10 males and 10 females, aged 20—50 years) who had prior experience consuming Makgeolli. The panelists were informed of the purpose, evaluation methods, and assessment criteria of the study, and preliminary training sessions using commercial Makgeolli samples were conducted once a week for three weeks to familiarize the panelists with the taste characteristics. The sensory evaluation was conducted in a sensory-evaluation room designed to minimize external stimuli. All samples were served in 200 mL glass cups containing 100 mL of Makgeolli, each coded with a random three-digit number to eliminate bias. The samples were presented in a randomized order, and the panelists were instructed to rinse their mouths with water between samples. The evaluation attributes included taste, color, aroma, and overall acceptability, assessed using a five-point hedonic scale (Herbert A & Joel LS 1993): 1=very poor, 2=poor, 3=fair, 4=good, and 5=very good.
6. Statistical Analysis
All physicochemical measurements were performed in triplicate (n=3), and the results are expressed as mean±standard deviation. Sensory evaluation was conducted by 20 panelists (n=20), and sensory scores are presented as mean±standard deviation. Statistical analysis was performed using a Statistics Package (Version 4.0; Analytical Software, Tucson, AZ, USA). Analysis of variance (ANOVA) was applied, and the significant differences among the means were determined using a Tukey’s multiple range test at p<0.05.
RESULTS AND DISCUSSION
1. Alcohol Content, pH, Titratable Acidity, and Soluble Solids (°Brix)
Table 1 lists the changes in alcohol content, pH, acidity, and Brix of Makgeolli prepared with JAE added at 0, 5, 10, 15, and 20% relative to rice. The ethanol content ranged from 6.08 to 6.12%, with no significant difference from the 6.10% measured in the control (0% JAE). The pH and titratable acidity also did not differ significantly, ranging from 4.02 to 4.07 and from 0.18 to 0.24% (w/v), respectively. The Brix value was 3.1 °Brix in the control and increased to 3.4 °Brix-4.2 °Brix with JAE addition, increasing with higher JAE supplementation. Previous studies on fermented rice beverages and products containing Jerusalem artichoke, such as Sikhye (Kim HS & Hwang ES 2021), jelly (Kim HS & Hwang ES 2022a), and Yakju (Choi JS et al 2023), reported that increasing the amount of Jerusalem artichoke leads to a higher solid content because of the sugars contained in the tuber, which is consistent with the present findings. On the other hand, no distinct differences in alcohol content were observed with JAE addition in this study. This result was attributed to the reduction in the amount of rice corresponding to the added JAE and the use of water-soluble inulin and sugars extracted from Jerusalem artichoke as fermentation substrates (Choi JS et al 2022). The yeast strain used in this study produces inulinase, an enzyme that hydrolyzes inulin to fermentable fructose (Choi JS et al 2022).
2. Color
Table 2 lists the Hunter color values (L*, a*, and b*) of Makgeolli fermented with 0%—20% JAE relative to rice. The L* value was 52.37 in the control Makgeolli without JAE, but decreased gradually to 40.00 as JAE supplementation was increased. The a* value, representing the range from red (+) to green (—), was —0.32 in the control, but increased from 1.11 to 3.21 proportionally with the JAE supplementation level. The b value was 6.72 in the control and ranged from 8.72 to 15.21 in the 5%—20% JAE-supplemented groups, showing a larger increase than the a value as the JAE supplementation level increased. Thus, Makgeolli without JAE showed a light white color, whereas increasing JAE addition resulted in a progressively darker reddish-yellow hue, a pattern observed in JAE itself and in the Makgeolli supplemented with JAE. This phenomenon was influenced by the discoloration of polyphenols contained in JAE (Kim YS et al 2011; Bach V et al 2013), the inherent pigments of Jerusalem artichoke, and the color generated during extraction by Maillard reactions. This color modification indicates a dark-colored Makgeolli, contrasting with the traditional white Makgeolli.
3. Mineral Content
Table 3 lists the mineral contents of Makgeolli supplemented with JAE. In both the control and JAE-supplemented samples (5%—20%), Na, K, Ca, Zn, Fe, Mg, Mn, and Cu were detected, whereas hazardous heavy metals, including As, Hg, Cd, and Pb, were not detected. In the control Makgeolli, the Na, K, and Ca contents were 145.11, 121.11, and 120.11 mg/L, respectively. The Na, K, and Ca concentrations at 20% JAE addition were 172.31, 146.29, and 140.22 mg/L, respectively. Wong B et al (2022) reported that the major minerals present in rice-based Makgeolli are P, Ca, K, Mg, and Zn, and the present results indicate that JAE supplementation can alter the mineral profile of Makgeolli by increasing several minerals.

Minerals content of Makgeolli with different concentration of Jerusalem artichoke water extract (mg/L)
In contrast, the Zn content was 80.29 mg/L in the control but decreased as JAE supplementation increased, reaching 32.00—35.18 mg/L at 15—20% supplementation. Because minerals were quantified by ICP-AES after nitric-acid digestion of filtered Makgeolli, the reported concentrations represent Zn retained in the filtrate after fermentation and filtration. The decline in Zn with increasing JAE may therefore reflect both formulation-related and fermentation-related factors. Jerusalem artichoke is rich in water-soluble carbohydrates such as inulin, while its Zn content has been reported to be relatively lower than that of rice, suggesting that partial substitution of rice-derived solids with JAE-derived components may lead to a net dilution of Zn in the final product volume (Impa SM & Johnson-Beebout SE 2012).
In addition, Zn is an essential trace mineral for yeast metabolism and contributes to enzymatic activity, cell growth, and fermentation performance (Walker GM 1988; Zhao H & Eide D 1996; Walker GM & Walker RS 2000). During fermentation, Zn can be actively taken up by yeast cells and become associated with microbial biomass and fermentation solids, which may subsequently be removed during filtration and thereby reduce Zn remaining in the liquid phase (Peña MM et al 1998; De Nicola R et al 2009). The increased availability of fermentable substrates from JAE may have promoted microbial activity and biomass formation, potentially enhancing Zn sequestration into fermentation solids. Furthermore, components present in JAE, including polyphenols and soluble dietary fiber, may interact with Zn and influence its distribution and bioaccessibility within the food matrix (Sandberg AS 2002; Hurrell RF 2003; Kim EY et al 2011; Ruta LL & Farcasanu IC 2020; Tokarczyk J et al 2025). Taken together, the Zn decrease observed with JAE supplementation is most appropriately interpreted as reduced Zn retention in the filtered liquid fraction due to combined effects of raw-material contribution and fermentation-associated redistribution.
Notably, fermentation performance indicators did not differ significantly among treatments, suggesting that Zn was not limiting under the present conditions. Future studies should quantify Zn in the raw materials and in spent lees to establish a Zn mass balance and should evaluate how filtration conditions, fermentation duration, and yeast-related factors affect Zn retention in the final product.
The Fe and Mg contents were 56.21 mg/L and 53.45 mg/L, respectively, in the control without JAE, but increased with JAE supplementation, reaching 89.71 mg/L and 79.77 mg/L, respectively, at the 20% addition level. The Mn and Cu contents in the control were 2.62 mg/L and 0.72 mg/L, respectively, in the control without JAE, and did not differ significantly with JAE supplementation.
4. Contents of Free Amino Acids and Their Derivatives
The contents of free amino acids and their derivatives in Makgeolli supplemented with JAE were analyzed, and Table 4 lists the results arranged in descending order. In the control without JAE, the contents of eight essential amino acids were in the following order: Phe > Lys > Trp > Val > Ile > Met > Thr > Leu > His, ranging from 9.00 to 47.11 mg/L. Their levels tended to increase at higher JAE addition. The total content of essential amino acids also increased with JAE supplementation, from 211.73 mg/L in the control to 235.14—254.07 mg/L in 5%—10% JAE, and 274.00—286.78 mg/L in 15%—20% JAE. The increase in essential amino acids contributes to the nutritional value of Makgeolli and plays an important role in the formation of characteristic fermented beverage flavors (Choi JS et al 2023). Leu and Val serve as precursors for higher alcohols and esters during fermentation, influencing the flavor of fermented beverages (Swiegers JH et al 2005), and are considered factors that enhance the flavor of JAE-supplemented Makgeolli. Among non-essential amino acids, Ala, Glu, and Arg showed relatively high contents, while Pro, Tyr, Gly, Cys, Asp, and Ser also tended to increase with JAE addition. In addition, the contents of L-ornithine, γ-aminobutyric acid, β-aminoisobutyric acid, cystathionine, ethanolamine, β-alanine, and L-α-aminoadipic acid were elevated. The total content of free amino acids and their derivatives increased from 942.23 mg/L in the control to 1,045.14—1,180.98 mg/L in the 5%—20% JAE treatments. The observed increase in functional amino acids, such as γ-aminobutyric acid, in JAE-supplemented Makgeolli, which is also detectable in control Makgeolli, suggests that they are produced by fermentation microorganisms, including yeast and lactic acid bacteria. Zhu L et al (2025) reported that γ-aminobutyric acid production is promoted by Saccharomyces cerevisiae. Among the lactic acid bacteria involved in fermentation, L. brevis, L. plantarum, and L. pentosus convert L-Glu into γ-aminobutyric acid via glutamate decarboxylase (Li H & Cao Y 2010). Furthermore, the increase in amino acid content with JAE addition indicates the effect of the Jerusalem artichoke itself. Therefore, the enhancement of free amino acids and their derivatives in Makgeolli supplemented with JAE significantly affects the flavor and functional properties (Bachmanov AA et al 2016; Choi JS et al 2023). Bakku RK et al (2022) reported a decrease in free amino acids in Yakju supplemented with raw Jerusalem artichoke, which was attributed to the Kunitz trypsin inhibitor present in Jerusalem artichoke (Bakku RK et al 2022). Nevertheless, the results differed from those of this study using the extract, suggesting that these inhibitory factors were inactivated during the JAE extraction process.
5. Total Polyphenol Content and Antioxidant Activity
Table 5 lists the total polyphenol content and antioxidant activities (DPPH and hydroxyl radical scavenging). The total polyphenol content of Makgeolli without JAE was 312.11 mg%, whereas it increased to 326.23—404.31 mg% with 5%—20% JAE supplementation, showing a trend of increasing content with higher JAE levels. The DPPH radical scavenging activity increased significantly from 23.62% in the control to 42.16%—64.11% in JAE-supplemented Makgeolli. The hydroxyl radical scavenging activity also significantly increased from 1.31% in the control to 2.11%—9.37% with increasing JAE addition. The JAE material used in this study contained comparable amounts of total polyphenols as reported by Kapusta I et al (2013), Showkat MM et al (2019), and Chen F et al (2014), and the observed antioxidant activity was attributed to these components (Kapusta I et al 2013; Chen F et al 2014; Showkat MM et al 2019). Nizioł-Łukaszewska Z et al (2018) also reported that a higher polyphenol content in the leaves and tubers of Jerusalem artichoke increased the DPPH and hydroxyl radical scavenging activities.
6. Sensory Characteristics
The sensory quality of Makgeolli supplemented with JAE was evaluated, and the results are listed in Table 6. The taste preference was higher for the JAE-supplemented samples than the control, and no significant difference was observed between the 15% and 20% addition levels. Although experiments with JAE additions above 20% were not conducted, the absence of a distinct taste difference between 15% and 20% suggests that the optimal Makgeolli was fermented with Jerusalem artichoke water extract (JAE) added at 0, 5, 10, addition level for taste is approximately 15%. The unique, slightly bitter flavor of Jerusalem artichoke, combined with effervescence, contributed to a distinctive taste. The color preference was also higher in the JAE-supplemented samples than in the control. The Makgeolli containing JAE showed a dark reddish-yellow hue, and the preference increased at higher addition levels. The flavor preference was higher than that of the control, but no significant difference was observed among the samples with 10%—20% JAE addition. This is likely because the flavor of Jerusalem artichoke resembles that of apple, almond, or walnut (Santis DD & Frangipane MT 2018) and may either dissipate during fermentation or blend with the alcohol flavor. The overall preference scores were high for samples with 5%—20% JAE addition, and the lack of significant differences between 10%—20% additions further supports 15% as the optimal addition level. Nevertheless, the sensory evaluation panel was somewhat limited in terms of consumer representativeness. Therefore, further studies will be needed to validate these findings.
SUMMARY AND CONCLUSION
Makgeolli was fermented with Jerusalem artichoke water extract (JAE) added at 0, 5, 10, 15, and 20% levels relative to rice to develop Makgeolli utilizing the functionality of Jerusalem artichoke. The physicochemical properties, mineral contents, free amino acids, and total polyphenol content with antioxidant activity were analyzed. The alcohol content ranged from 6.08% to 6.12%, showing no significant difference from the control (6.10%). The pH and titratable acidity were 4.02-4.07 and 0.18—0.24% (w/v), respectively, with no notable differences. The soluble solids (°Brix) were slightly higher in the JAE-supplemented samples (3.4%—4.2%) compared to the control (3.1%). In terms of the Hunter color, L* decreased from 52.37 for the control to 40.00 for up to 20% JAE addition; a* increased from —0.32 in the control to 3.21 in 20% additions, and b* exhibited a larger increase than a*. The Na, K, and Ca contents ranged from 145.11—172.31 mg/L, 121.11—146.29 mg/L, and 120.11—146.29 mg/L with JAE 0%—20% addition. Zn decreased from 80.29 mg/L in the control to 32.00 mg/L with 15% JAE and 35.18 mg/L with 20% JAE. Fe and Mg increased with JAE addition, reaching 89.71 mg/L and 79.77 mg/L at 20% JAE, respectively, compared to 56.21 mg/L and 53.45 mg/L in the control. The Mn and Cu contents were 2.62 mg/L and 0.72 mg/L in the control, showing no significant difference with JAE addition. Toxic elements, such as As, Hg, Cd, and Pb, were not detected. The essential amino acid contents (mg/L) increased with JAE addition in the order of Phe (47.11) > Lys (46.17) > Trp (33.90) > Val (33.29) > Ile (17.12) > Met (13.03) > Thr (12.11) > Leu (10.62) > His (9.00). Among the free amino acids, Ala, Glu, and Arg were abundant, and Pro, Tyr, Gly, Cys, Asp, and Ser also tended to increase with JAE addition. Functional amino acids such as L-ornithine and γ-aminobutyric acid were detected and increased with JAE addition. The total polyphenol content increased from 312.11 mg% in the control to 326.23—404.31 mg% with 5%—20% JAE addition. DPPH radical scavenging activity also increased from 23.62% in the control to 42.16%—64.11% in the JAE-supplemented samples, and hydroxyl radical scavenging activity increased at higher JAE contents. The sensory evaluation of taste, color, flavor, and overall preference indicated that the optimal JAE addition level was approximately 15%.
Acknowledgments
This research was supported by the Regional Innovation System & Education (RISE) Program, specifically the Regional Growth Innovation Lab, through the Gyeongbuk RISE Center, Funded by the Ministry of Education (MOE) and Gyeongsangbuk-do, Republic of Korea (2025-RISE-15-205).
References
-
Bach V, Jensen S, Clausen MR, Bertram HC, Edelenbos M (2013) Enzymatic browning and after‑cooking darkening of Jerusalem artichoke tubers (Helianthus tuberosus L). Food Chem 141(2): 1445-1450.
[https://doi.org/10.1016/j.foodchem.2013.04.028]

-
Bachmanov AA, Bosak NP, Glendinning JI, Inoue M, Li X, Manita S, McCaughey SA, Murata Y, Reed DR, Tordoff MG, Beauchamp GK (2016) Genetics of amino acid taste and appetite. Adv Nutr 7(4): 806S-822S.
[https://doi.org/10.3945/an.115.011270]

-
Bakku RK, Gupta R, Min CW, Kim ST, Takahashi G, Shibato J, Shioda S, Takenoya F, Agrawal GK, Rakwal R (2022) Unravelling the Helianthus tuberosus L (Jerusalem artichoke, Kiku-Imo) tuber proteome by label-free quantitative proteomics. Molecules 27(3): 1111.
[https://doi.org/10.3390/molecules27031111]

-
Blois MS (1958) Antioxidant determination by the use of a stable free radical. Nature 181: 1199-1200.
[https://doi.org/10.1038/1811199a0]

-
Chen F, Long X, Liu Z, Shao H, Liu L (2014) Analysis of phenolic acids of Jerusalem artichoke (Helianthus tuberosus L) Responding to salt-stress by liquid chromatography/tandem mass spectrometry. Scientific World Journal 2014: 568043
[https://doi.org/10.1155/2014/568043]

-
Chi Z, Chi Z, Zhang T, Liu G, Yue L (2009) Inulinase-expressing microorganisms and applications of inulinases. Appl Microbiol Biotechnol 82(2): 211-220.
[https://doi.org/10.1007/s00253-008-1827-1]

-
Choi JS, Choi KT, Kim CW, Lee SB, Park HD (2022) Fermentation characteristics of Yakju supplemented with Jerusalem artichoke (Helianthus tuberosus L) and co-fermented by inulin‑degrading yeast Wickerhamomyces anomalus JS01 and Saccharomyces cerevisiae KMBL7001. Food Sci Preserv 29(5): 800-812.
[https://doi.org/10.11002/kjfp.2022.29.5.800]

-
Choi JS, Choi KT, Kim CW, Park HD, Lee SB (2023) Fermentation characteristics of Yakju containing different amounts of steam-cooked Jerusalem artichoke (Helianthus tuberosus L). Food Sci Preserv 30(1): 155-169.
[https://doi.org/10.11002/kjfp.2023.30.1.155]

-
Chung SK, Osawa T, Kawakishi S (1997) Hydroxyl radical‑scavenging effects of spices and scavengers from brown mustard (Brassica nigra). Biosci Biotechnol Biochem 61(1): 118-123.
[https://doi.org/10.1271/bbb.61.118]

- De Nicola R, Hall N, Bollag T, Thermogiannis G, Walker GM (2009). Zinc accumulation and utilization by wine yeasts. Int J Wine Research 1: 85-94.
-
Guiraud JP, Bajon AM, Chautard P, Galzy P (1983) Inulin hydrolysis by an immobilized yeast-cell reactor. Enzyme Microb Technol 5(3): 185-190.
[https://doi.org/10.1016/0141-0229(83)90092-3]

-
Guo X, Xie Z, Wang G, Zou Q, Tang R (2018) Effect on nutritional, sensory, textural and microbiological properties of low-fat yogurt supplemented with Jerusalem artichoke powder. Int J Dairy Technol 71(S1): 167-174.
[https://doi.org/10.1111/1471-0307.12404]

- Herbert A, Joel LS (1993) In Sensory Evaluation Practices. 2nd ed. Academic Press, NY. USA. pp 68-94.
-
Hurrell RF (2003) Influence of vegetable protein sources on trace element and mineral bioavailability. J Nutr 133(9): 2973S-2977S.
[https://doi.org/10.1093/jn/133.9.2973S]

-
Impa SM, Johnson-Beebout SE (2012) Mitigating zinc deficiency and achieving high grain Zn in rice through integration of soil chemistry and plant physiology research. Plant Soil 361: 3-41.
[https://doi.org/10.1007/s11104-012-1315-3]

- Kapusta I, Krok ES, Jamro DB, Cebulak T, Kaszuba J, Salach RT(2013) Identification and quantification of phenolic compounds from Jerusalem artichoke (Helianthus tuberosus L) tubers. J Food Agric Environ 11(3): 601-606.
-
Khatun MM, Liu CG, Zhao XQ, Yuan WJ, Bai FW (2017) Consolidated ethanol production from Jerusalem artichoke tubers at elevated temperature by Saccharomyces cerevisiae engineered with inulinase expression through cell surface display. J Ind Microbiol Biotechnol 44(2): 295-301.
[https://doi.org/10.1007/s10295-016-1881-0]

-
Kim EY, Pai TK, Han O (2011) Effect of bioactive dietary polyphenols on zinc transport across the intestinal Caco-2 cell monolayers. J Agric Food Chem 59(8): 3606-3612.
[https://doi.org/10.1021/jf104260j]

-
Kim HS, Hwang ES (2021) Quality characteristics and antioxidant activities of ‘Sikhye’ containing different amounts of Jerusalem artichoke extract. Food Sci Preserv 28(6): 771-779
[https://doi.org/10.11002/kjfp.2021.28.6.771]

-
Kim HS, Hwang ES (2022a) Quality characteristics and antioxidant activity of jelly containing varying amounts of Jerusalem artichoke extract. J Korean Soc Food Sci Nutr 51(5): 476-482.
[https://doi.org/10.3746/jkfn.2022.51.5.476]

-
Kim HS, Hwang ES (2022b) Quality characteristics and antioxidant activity of brown rice porridge supplemented with Jerusalem artichoke (Helianthus tuberosus L). J Korean Soc Food Sci Nutr 51(4): 352-359.
[https://doi.org/10.3746/jkfn.2022.51.4.352]

-
Kim YS, Lee SJ Lee, Woo HJ, Kim EH, Park PJ, Tae JB (2011). Antioxidant activity and protective effects of extracts from Helianthus tuberosus L leaves on t-BHP induced oxidative stress in chang cells. J Korean Soc Food Sci Nutr 40(11): 1525-1531.
[https://doi.org/10.3746/jkfn.2011.40.11.1525]

- Lee EH, Lee YJ, Choi OB, Kang SM (2007) Effect of a combined diet of Jerusalem artichoke’s inulin, lotus leaf and herb extracts in obesity-induced white rat with fat diet. J Korean Soc Appl Biol Chem 50(4): 295-303.
-
Li H, Cao Y (2010) Lactic acid bacterial cell factories for γ-aminobutyric acid. Amino Acids 39(5): 1107-1116.
[https://doi.org/10.1007/s00726-010-0582-7]

-
Li J, Jia S, Yuan C, Yu B, Zhang Z, Zhao M, Liu P, Li X, Cui B (2022) Jerusalem artichoke inulin supplementation ameliorates hepatic lipid metabolism in type 2 diabetes mellitus mice by modulating the gut microbiota and fecal metabolome. Food Func 13(22): 11503-11517.
[https://doi.org/10.1039/D2FO02051C]

-
Lim SH, Ryu JM, Lee H, Jeon JH, Sok DE, Choi ES (2010) Ethanol fermentation from Jerusalem artichoke powder using Saccharomyces cerevisiae KCCM50549 without pretreatment for inulin hydrolysis. Bioresour Technol 102(2): 2109-2111.
[https://doi.org/10.1016/j.biortech.2010.08.044]

-
Liu ZX, Steinberger Y, Chen X, Wang JS, Xie GH (2015) Chemical composition and potential ethanol yield of Jerusalem artichoke in a semi-arid region of China. Ital J Agron 10(1): 603.
[https://doi.org/10.4081/ija.2015.603]

-
Nair KK, Kharb S, Thompkinson DK (2010) Inulin dietary fiber with functional and health attributes (a review). Food Rev Int 26(2): 189-203.
[https://doi.org/10.1080/87559121003590664]

-
Nizioł-Łukaszewska Z, Furman-Toczek D, Zagórska-Dziok M (2018) Antioxidant activity and cytotoxicity of Helianthus tuberosus tubers and leaves extract. Lipids Health Dis 17(1): 280.
[https://doi.org/10.1186/s12944-018-0929-8]

-
Park HS (2010) Quality characteristics of Sulgidduk by the addition of Jerusalem artichoke (Helianthus tuberosus L.) powder. Culi Sci & Hos Res 16(3): 259-267.
[https://doi.org/10.20878/cshr.2010.16.3.018]

-
Peña MM, Koch KA, Thiele DJ (1998) Dynamic regulation of copper and zinc uptake in Saccharomyces cerevisiae. Mol Cell Biol 18(5): 2514-2523.
[https://doi.org/10.1128/MCB.18.5.2514]

-
Ruta LL, Farcasanu IC (2020) Interaction between polyphenolic antioxidants and Saccharomyces cerevisiae cells defective in heavy metal transport across the plasma membrane. Biomolecules 10(11): 1512.
[https://doi.org/10.3390/biom10111512]

-
Sandberg AS (2002) Bioavailability of minerals in legumes. Br J Nutr 88(S3): S281-S285.
[https://doi.org/10.1079/BJN/2002718]

-
Santis DD, Frangipane MT (2018) Evaluation of chemical composition and sensory profile in Jerusalem artichoke (Helianthus tuberosus L) tubers: The effect of clones and cooking conditions. Int J Gastron Food Sci 11: 25-30.
[https://doi.org/10.1016/j.ijgfs.2017.11.001]

-
Sawicka B, Skiba D, Pszczółkowski P, Aslan I, Sharifi-Rad J, Krochmal-Marczak B (2020) Jerusalem artichoke (Helianthus tuberosus L) as a medicinal plant and its natural products. Cell Mol Biol (Noisy-le-grand) 66(4): 160-177.
[https://doi.org/10.14715/cmb/2020.66.4.20]

-
Shao T, Yu Q, Zhu T, Liu A, Gao X, Long X, Liu Z (2020) Inulin from Jerusalem artichoke tubers alleviates hyperglycaemia in high-fat-diet-induced diabetes mice through the intestinal microflora improvement. Br J Nutr 123(3): 308-318
[https://doi.org/10.1017/S0007114519002332]

-
Sheng J, Chi ZM, Li J, Gao L, Gong F (2007) Inulinase production by the marine yeast Cryptococcus aureus G7a and inulin hydrolysis by the crude inulinase. Process Biochem 42(5): 805-811.
[https://doi.org/10.1016/j.procbio.2007.01.016]

-
Shin MH, Chung NY (2019) Quality evaluation of brown rice Sulgidduk added with Jerusalem artichoke powder. J East Asian Soc Diet Life 29(2): 112-119.
[https://doi.org/10.17495/easdl.2019.4.29.2.112]

- Shin SH, Kwon SJ, Jo HJ, Go D, Han J (2012) Extraction and analysis of inulin from Jerusalem artichoke. Food Sci Ind 45(4): 50-58.
-
Showkat MM, Falck-Ytter AB, Strætkvern KO (2019) Phenolic acids in Jerusalem artichoke (Helianthus tuberosus L): Plant organ dependent antioxidant activity and optimized extraction from leaves. Molecules 24(18): 3296.
[https://doi.org/10.3390/molecules24183296]

-
Singleton VL, Orthofer R, Lamuela-Raventós RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol 299: 152-178.
[https://doi.org/10.1016/S0076-6879(99)99017-1]

- Skujins S (1998) Handbook for ICP-AES (Varian-Vista). A Short Guide to Vista Series. ICP-AES Operation, Version 1.0; Varian Int. AG, Zug, Switzerland.
-
Swiegers JH, Bartowsky EJ, Henschke PA, Pretorius IS (2005) Yeast and bacterial modulation of wine aroma and flavour. Aus J Grape Wine Res 11(2): 139-173.
[https://doi.org/10.1111/j.1755-0238.2005.tb00285.x]

-
Tokarczyk J, Jaworowska A, Kowalczyk D, Kasprzak M, Jagielski P, Koch W (2025) Influence of diet on the bioaccessibility of Zn from dietary supplements: Findings from an in vitro digestion model and analytical determinations. Nutrients 18(1): 94.
[https://doi.org/10.3390/nu18010094]

-
Vyacheslavov AV, Bichaev VB, Titova AD, Rybin DS, Ermolaeva TN (2018) ICP-AES Analysis of secondary tungsten-containing raw materials for production of hard alloys. Inorg Mater 54(14): 1430-1434.
[https://doi.org/10.1134/S0020168518140169]

- Walker GM (1998) Yeast Physiology and Biotechnology. John Wiley & Sons, Chichester, USA. pp 203-210.
- Walker GM, Walker RS (2000) Enhancing yeast alcoholic fermentations. Adv Appl Microbiol 105: 87-129.
-
Wang YZ, Zou SM, He ML, Wang CH (2015) Bioethanol production from the dry powder of Jerusalem artichoke tubers by recombinant Saccharomyces cerevisiae in simultaneous saccharification and fermentation. J Ind Microbiol Biotechnol 42(4): 543-551.
[https://doi.org/10.1007/s10295-014-1572-7]

-
Wong B, Muchangi K, Quach E, Chen T, Owens A, Otter D, Phillips M, Kam R (2022) Characterisation of Korean rice wine (Makgeolli) prepared by different processing methods. Curr Res Food Sci 6: 100420.
[https://doi.org/10.1016/j.crfs.2022.100420]

-
Zhao H, Eide D (1996) The yeast zrt1 gene encodes the zinc transporter protein a high-affinity uptake system induced by zinc limitation. Proc Natl Acad Sci USA 93(6): 2454-2458.
[https://doi.org/10.1073/pnas.93.6.2454]

-
Zhu L, Peng Y, Wen B, Liu X, Yang W, Xu W, Li M, Zhang J, Liu J (2025) Process optimization of GABA enrichment and sensory flavor improvement in Huangjinya green tea through Saccharomyces cerevisiae fermentation. LWT-Food Sci Technol 236: 118707.
[https://doi.org/10.1016/j.lwt.2025.118707]

