Effect of High Temperature and High Pressure on Antioxidant Activity of Cultivated Wild Ginseng
Abstract
This study investigated the impact of high-temperature and high-pressure (HTHP) steaming on the antioxidant properties of cultivated wild ginseng. Steaming was performed at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²). The total phenolic and flavonoid contents and antioxidant capacities of the resulting extracts were analyzed using 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), ferric reducing antioxidant power (FRAP), oxygen radical absorbance capacity (ORAC), and hydroxyl radical scavenging assays. Higher temperatures and pressures resulted in higher total phenolic (6.04 mg GAE/g) and flavonoid (2.11 mg QE/g) contents. The antioxidant activities also showed significant enhancement (p<0.05) under elevated processing conditions, reaching maximum values for DPPH (14.42 mM TE/g), ABTS (68.94 mM TE/g), FRAP (675.75 μM TE/g), ORAC (265.43 mg TE/g), and hydroxyl radical scavenging activity (34.40%) at 132℃ (2.0 kgf/cm²). These improvements were attributed to the thermal degradation and transformation of phenolic compounds, the formation of low-molecular-weight antioxidants, and the generation of Maillard reaction products, such as melanoidins, with strong reducing and radical scavenging abilities. Overall, HTHP steaming enhanced the antioxidant potential of cultivated wild ginseng significantly, suggesting that treatment at 132℃ (2.0 kgf/cm²) is optimal for maximizing the bioactivity and may contribute to the development of functional ginseng products with improved health-promoting properties.
Keywords:
high-temperature, high-pressure, cultivated wild ginseng, antioxidant activityINTRODUCTION
Ginseng (Panax ginseng C.A. Meyer), belonging to the Araliaceae family, has been widely utilized as a natural medicinal resource in East Asia for thousands of years, particularly in Korea, China, and Japan (Yun TK 2001). The term “Panax,” derived from the Greek words Pan (all) and Axos (cure), signifies “a cure for all diseases” (Hu SY 1976). Traditionally, ginseng roots have been used in Korean and Chinese medicine and are increasingly recognized in Western countries for their tonic, restorative, and therapeutic properties (Kim JH & Nah SY 2005). Among various types, cultivated wild ginseng is found in natural mountainous areas, while cultivated wild ginseng is artificially grown in forest environments under natural conditions. Cultivated wild ginseng is considered superior to regular cultivated ginseng because it contains higher levels of certain bioactive components, particularly ginsenosides, despite slower growth rates (Lim W et al 2005). The major bioactive constituents of ginseng include saponins, primarily ginsenosides, and nonsaponin compounds such as panacen, polyacetylene derivatives, and phenolic compounds (Gillis CN 1997).
Traditionally, ginseng has been consumed in three primary forms: fresh ginseng (susam), white ginseng (baeksam, dried ginseng), and red ginseng (hongsam). Secondary processed forms, such as ginseng concentrates, were historically limited. However, recent technological advancements have led to the development of various novel ginseng products, including “sun ginseng,” “puffed red ginseng,” and “black ginseng,” designed to enhance bioactive compound profiles and improve functional efficacy. Among the various processing techniques, thermal treatment remains one of the most commonly employed due to its ability to alter the chemical composition, improve pharmacological properties, and enhance sensory attributes such as taste, aroma, and color (Lee JH et al 2006). Heat treatment in the processing of food and medicinal materials aims to reduce toxicity, extend shelf life, and enhance therapeutic properties. Nevertheless, it can also result in nutrient degradation and the loss of thermolabile bioactive substances. Interestingly, several studies have demonstrated that high-temperature and high-pressure (HTHP) treatment can increase polyphenolic and flavonoid contents, thereby enhancing antioxidant capacity in a variety of food sources, including shiitake mushrooms (Choi Y et al 2006), licorice (Woo KS et al 2006), garlic (Kwon OC et al 2006), pear (Hwang IG et al 2006), ginseng (Yang SJ et al 2006), tomato (Dewanto V et al 2002), and citrus peel (Jeong SM et al 2004). These findings suggest that chemical transformations induced by thermal processing may contribute to elevated bioactive compound levels and improved functional properties.
Red ginseng, which constitutes a major portion of commercial ginseng products, is produced by steaming fresh ginseng roots for a specific duration. This process promotes the formation of unique compounds, such as maltol, and ginsenosides characteristic of red ginseng, including Rg3. Heat treatment significantly increases the total phenolic and flavonoid contents, both known for their antioxidant activity, while also contributing to color enhancement through browning reactions and the accumulation of free sugars (Yang SJ et al 2006; Hong HD et al 2007; Kwak YS et al 2008). Recent research has focused on refining ginseng processing to develop products with superior biofunctional properties. For instance, “sun ginseng,” produced by steaming ginseng at approximately 120℃, demonstrates increased antioxidant and anticancer activities (Kim WY et al 2000; Park JH 2004). Similarly, “black ginseng,” obtained through repeated steaming and drying cycles, exhibits enhanced antioxidant and anticancer efficacy (Jo HK et al 2011; Nam KY et al 2012). Therefore, the present study aimed to establish a rapid high-temperature and high-pressure (HTHP) processing method to enhance the functional properties of cultivated wild ginseng. By evaluating the variations in antioxidant activity under different HTHP conditions, this study provides fundamental insights into the potential utilization of cultivated wild ginseng as a high-value functional material and supports the development of novel ginseng-based products.
MATERIALS AND METHODS
1. Materials and Reagents
Five-year-old cultivated wild ginseng roots were collected (August 5, 2024) from the experimental field of Jinsaengbio Farm Association Co. (Hamyang-gun, Gyeongnam, Korea). Folin-Ciocalteu reagent, sodium carbonate, gallic acid, aluminum chloride, quercetin, 2,2-diphenyl-1-picrylhydrazyl (DPPH), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), 2,2-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), 2,2-azobis(2-methylpropionamidine) dihydrochloide (AAPH), potassium dihydrogen phosphate (KH2PO4), sodium phosphate dibasic dodecahydrate (Na2HPO4·12H2O), iron(III) chloride hexahydrate (FeCl3·6H2O), 2,4,6-tripyridyl-s-triazine (TPTZ), and p-nitrosodimethylaniline (p-NDA) were purchased from Sigma-Aldrich. All other chemicals used in the experiments were of analytical grade, and deionized water was used throughout.
2. Sample Preparation
The main roots of cultivated wild ginseng were thoroughly washed with tap water to remove soil and debris. The first-dried cultivated wild ginseng was prepared by drying fresh roots in a forced-convection oven at 40℃ for 6 hrs until the moisture content was reduced to below 30%. The partially dried samples were then steamed at 95℃ for 1.5 hrs using an autoclave (HK-AC60, Hankuk S&I Co., Korea). After cooling to room temperature, the steamed ginseng was subjected to secondary drying at 60℃ for 3 hrs in a forced-convection drying oven. Subsequently, the twice-dried ginseng samples were steamed again under different temperature and pressure conditions—95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²)—using an autoclave, followed by drying at 60℃ for 3 hrs to obtain the temperature-treated cultivated wild ginseng. And then the steamed cultivated wild ginseng was finely ground, and the main root and fine root fractions were blended at a weight ratio of 8.5:1.5. Ten grams of the homogenized sample was placed into the extraction cell of an accelerated solvent extraction system (Dionex ASE 350, Thermo Scientific, Idstein, Germany). Extraction was conducted under optimal ginsenoside extraction conditions determined in preliminary experiments, using 88% ethanol as the solvent at 142℃ for 29 mins. The resulting extract was concentrated using either a multi-evaporator (Multivapor P-6, Flawil, Switzerland) or a rotary evaporator (SB-1200, Eyela, Tokyo, Japan) to remove ethanol at 40℃. The concentrated extract was frozen at —75℃ for 24 hrs in a deep freezer (DF8514, ILSHIN Biobase Co., Korea) and subsequently freeze-dried using a freeze dryer (FDU-1200, Eyela, Tokyo, Japan). The lyophilized extract was finely pulverized using a mortar and pestle and stored at —75℃ until further analysis.
3. Determination of Total Phenolic Content
The total phenolic content of each extract was colorimetrically estimated using the Folin-Ciocalteu method (Singleton VL & Rossi JA 1965). More specifically, a portion a 1:9 (v/v) mixture of Folin-Ciocalteu reagent (2.5 mL) and distilled water was added to each extract (0.5 mL, concentration 20 mg/mL). The resulting mixtures were allowed to stand at room temperature for 5 min prior to the addition of an aqueous 7.5% Na2CO3 solution (2 mL). The obtained solutions were then allowed to stand at room temperature for a further 1 h, after which time their absorbances at 765 nm using a spectrophotometer (Synergy HTX; Biotech Instruments., Winooski, VT, USA) were measured using gallic acid and methanol as the reference standard and blank solutions, respectively. All values were expressed as mg of gallic acid equivalents (GAE) per g of the steamed cultivated wild ginseng extracts.
4. Determination of Total Flavonoid Content
Total flavonoid content was determined using a colourimetric method described (Woisky RG & Salatino A 1998). A 0.5 mL of each extract (concentration 20 mg/mL) was taken and 0.5 mL of 2% ethanolic solution of AlCl3 was added to it. After 1 h at room temperature, the absorbance was read at 420 nm using a spectrophotometer (Synergy HTX; Biotech Instruments., Winooski, VT, USA). All values were expressed as mg quercetin equivalents (QE) per g of the steamed cultivated wild ginseng extracts.
5. DPPH Radical Scavenging Activity
The DPPH radical scavenging activity of the steamed cultivated wild ginseng extracts was evaluated using the method of Delgado-Andrade C et al (2005). A 200 μL aliquot of each complex was added to 1 mL of DPPH solution (74 mg/L in methanol). The DPPH solution, prepared fresh daily, had an initial absorbance of 1.8 at 520 nm. After mixing, the solution was shaken for 30 minutes, and absorbance was measured at 520 nm. Trolox solutions (0.05—0.5 mM) were used to generate a standard calibration curve. All measurements were performed in quintuplicate, and results were expressed as mM Trolox equivalents (TE) per gram of the steamed cultivated wild ginseng extracts.
6. ABTS Radical Scavenging Activity
The ABTS radical scavenging capacity was assessed according to the method of Re R et al (1999). ABTS radical cation (ABTS⁺·) was generated by reacting a 7 mM ABTS stock solution with 2.45 mM potassium persulfate (K2S2O8), followed by dilution with 5 mM PBS (pH 7.4) to achieve an absorbance of 0.70±0.02 at 730 nm. To 3 mL of the ABTS⁺·solution, 50 μL of each sample or Trolox standard was added. Absorbance was recorded at 730 nm for 20 minutes using a spectrophotometer (Synergy HTX spectrophotometer, Biotech Instruments, Winooski, VT, USA). Trolox standards were used for calibration. All measurements were conducted five times, and results were expressed as mM TE/g of the steamed cultivated wild ginseng extracts.
7. Ferric Reducing Antioxidant Power (FRAP)
The ferric-reducing ability was evaluated according to the method of Benzie IF & Strain JJ (1996). A reaction mixture consisting of 900 μL of FRAP reagent, 90 μL of distilled water, and 50 μL of each sample was prepared. The FRAP reagent was freshly prepared by mixing 2.5 mL of 10 mM TPTZ in 40 mM HCl, 2.5 mL of 20 mM FeCl3·6H2O, and 25 mL of 0.3 M acetate buffer (pH 3.6). The absorbance was monitored every 15 seconds for 30 minutes at 595 nm and 37℃ using a spectrophotometer (Synergy HTX spectrophotometer, Biotech Instruments, Winooski, VT, USA). Trolox solutions were used for calibration. Each sample was measured five times, and results were expressed as μM TE/g of the steamed cultivated wild ginseng extracts.
8. Oxygen Radical Absorbance Capacity Activity (ORAC)
The ORAC assay was conducted as described by Huang D et al (2002). A 153 mM solution of 2,2-azobis(2-methylpropionamidine) dihydrochloide (AAPH) was prepared fresh daily in 75 mM phosphate buffer (pH 7.4). A fluorescein stock solution (4 × 10⁻³ mM) was diluted 1:1000 in the same buffer. In a microplate, 150 μL of the fluorescein working solution was added to each interior well, followed by 25 μL of phosphate buffer (blank), Trolox standard, or sample. Plates were equilibrated at 37℃ for 30 minutes. The reaction was initiated by adding 25 μL of AAPH using a microplate reader injector. Fluorescence was measured every minute for 90 minutes at an excitation of 485 nm and emission of 520 nm. Each sample was analyzed five times. ORAC values were calculated based on area under the curve (AUC) as follows:
Where R1 is the fluorescence reading at the initiation of the reaction and Rn is the last measurement.
9. Hydroxyl Radical Scavenging Activity by p-NDA
Hydroxyl radical scavenging activity was determined using the p-nitrosodimethylaniline (p-NDA) assay described by Srinivasan R et al (2007). A reaction mixture containing 0.5 mL of each sample in DMSO was mixed with 0.5 mL each of 0.1 mM ferric chloride, 0.1 mM EDTA, 0.1 mM ascorbic acid, 2 mM hydrogen peroxide, and 0.01 mM p-NDA in 20 mM phosphate buffer (pH 7.4). The final volume was 3 mL. Absorbance was measured at 440 nm. All samples were measured five times.
10. Statistical Analysis
The experimental data were subjected to analysis of variance (ANOVA). Significant differences between the mean values, as determined from measurements carried out in five replicate tests (i.e., p<0.05), were obtained by Duncan’s multiple-range test using statistical analysis software (SPSS 20.0, IBM Inc., NY, USA).
RESULTS AND DISCUSSION
1. Total Phenolic Content
The total phenolic content of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure steaming treatments at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) is presented in Table 1. The total phenolic content was measured as 4.71±0.01 mg GAE/g at 95℃ (0 kgf/cm²), 4.95±0.01 mg GAE/g at 117℃ (1.0 kgf/cm²), 5.90±0.01 mg GAE/g at 126℃ (1.5 kgf/cm²), and 6.04±0.02 mg GAE/g at 132℃ (2.0 kgf/cm²), demonstrating a gradual increase in total polyphenol content with increasing temperature and pressure (p<0.05). Similarly, Yang SJ et al (2006) reported that the total phenolic content of fresh ginseng (2.68 mg GAE/g) increased significantly to 3.93—29.46 mg GAE/g following high-temperature and high-pressure thermal processing, with higher temperatures and longer heating durations leading to greater phenolic content. These results are consistent with the findings of the present study. The observed enhancement in total phenolic compounds following heat treatment is likely due to the conversion of bound phenolic compounds into free phenolic forms, which are more readily extractable, or to the thermal degradation of high-molecular-weight polyphenols into low-molecular-weight phenolic compounds.
2. Total Flavonoid Content
The total flavonoid content of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure steaming at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) is summarized in Table 2. The flavonoid content was measured as 1.66±0.01, 1.70±0.01, 1.96±0.01, and 2.11±0.01 mg QE/g at 95℃, 117℃, 126℃, and 132℃, respectively, with the highest value observed at 132℃ (2.0 kgf/cm²) (p<0.05). These results indicate a positive correlation between processing temperature and pressure and the total flavonoid content. Consistent with previous findings by Yang SJ et al (2006), where fresh ginseng exhibited a flavonoid content of 0.39 mg/g that increased to 0.42—4.75 mg/g after high-temperature and high-pressure treatment, the current study also demonstrates an increase in flavonoid content with higher pressure and prolonged heating, mirroring the trend observed for total phenolic compounds.
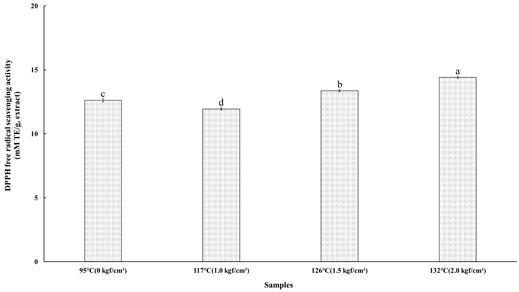
3. DPPH Radical Scavenging Activity
Fig. 1 presents the DPPH radical scavenging activity of the steamed cultivated wild ginseng extracts following high-temperature and high-pressure steaming at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²). The activities were determined to be 12.62±0.12, 11.94±0.09, 13.37±0.08, and 14.42±0.09 mM TE/g at 95℃, 117℃, 126℃, and 132℃, respectively. Statistically significant differences were observed among the treatment conditions (p<0.05), with the DPPH radical scavenging activity exhibiting the following order: 132℃ (2.0 kgf/cm²) > 126℃ (1.5 kgf/cm²) > 95℃ (0 kgf/cm²) > 117℃ (1.0 kgf/cm²). These results indicate that DPPH radical scavenging activity is enhanced by increasing temperature and pressure, reaching a maximum at 132℃ (2.0 kgf/cm²). Previous studies have demonstrated that phenolic compounds play a pivotal role in the formation of melanoidins, with a portion of these compounds remaining integrated within their structure. Patrignani M & González-Forte LdS (2021) reported that the antioxidant activity of melanoidins is augmented due to phenolic compounds binding to the carbohydrate backbone generated during the Maillard reaction. Similarly, Tores de la Cruz S et al (2019) suggested that during coffee roasting, phenolic compounds can associate with the initial melanoidin skeleton composed of carbohydrates and proteins. Given their well-established strong antioxidant properties, phenolic compounds likely act synergistically with melanoidins in the gastrointestinal tract (Saura-Calixto F 2011).
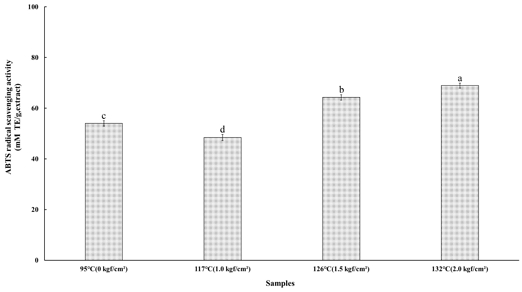
4. ABTS Radical Scavenging Activity
When ABTS and hydrogen peroxide are left to react in the dark, ABTS radicals are generated through a chemical reaction. These ABTS radicals are reduced by the active hydroxyl (-OH) groups of antioxidant compounds, resulting in the decolorization of the characteristic blue-green color of the radical. This decrease in absorbance is used to quantify the antioxidant activity of the extract. The ABTS radical scavenging assay is a widely used and rapid method for evaluating antioxidant capacity (Ko HM et al 2018). The ABTS radical scavenging activity of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure steaming at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) is presented in Fig. 2. The ABTS radical scavenging capacities were 53.99±1.04 mM TE/g at 95℃, 48.42±1.20 mM TE/g at 117℃, 64.24±1.04 mM TE/g at 126℃, and 68.94±0.98 mM TE/g at 132℃. A significant difference was observed among treatments (p<0.05), with antioxidant activity increasing in the order of 132℃ (2.0 kgf/cm²) > 126℃ (1.5 kgf/cm²) > 95℃ (0 kgf/cm²) > 117℃ (1.0 kgf/cm²). The highest ABTS radical scavenging activity was observed under the 132℃ (2.0 kgf/cm²) condition. This result aligns with the findings of Kim JS & Lee YS (2009), who reported higher ABTS values than DPPH in antioxidant assays involving melanoidins formed from various isomers. The difference between TEAC and DPPH assay results may be attributed to variations in reaction media (aqueous for ABTS and methanolic for DPPH) and their distinct mechanisms of action. While both DPPH· and ABTS+· radicals primarily undergo electron transfer reactions, DPPH may additionally exhibit minor hydrogen atom transfer behavior (Ichikawa K et al 2019).
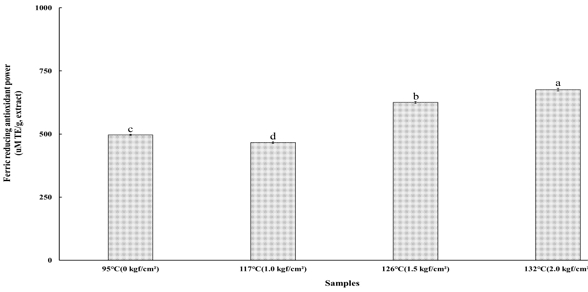
5. Ferric Reducing Antioxidant Power
The ferric reducing antioxidant power (FRAP) assay measures the ability of antioxidants to reduce ferric (Fe³⁺) to ferrous (Fe²⁺) ions, based on their reduction potentials relative to the Fe³⁺/Fe²⁺ redox couple (Huang D et al 2005). In this assay, the chromogenic ligand 2,4,6-tripyridyl-s-triazine (TPTZ) forms a blue-colored complex specifically with Fe²⁺. When antioxidants in the sample donate electrons to Fe³⁺, it is reduced to Fe²⁺, which subsequently reacts with TPTZ to produce a stable Fe²⁺—TPTZ complex. The resulting increase in absorbance at approximately 595 nm is directly proportional to the total reducing power of the antioxidants present in the sample (Benzie IFF & Szeto YT 1999). The FRAP values of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure (HTHP) steaming treatments at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) are shown in Fig. 3. The FRAP values were 497.06±2.00 μM TE/g at 95℃, 466.39±3.33 μM TE/g at 117℃, 625.08±4.00 μM TE/g at 126℃, and 675.75±5.33 μM TE/g at 132℃. A significant difference was observed among the treatment groups (p<0.05), with antioxidant reducing capacity increasing in the order of 132℃ (2.0 kgf/cm²) > 126℃ (1.5 kgf/cm²) > 95℃ (0 kgf/cm²) > 117℃ (1.0 kgf/cm²). The highest FRAP value observed at 132℃ (2.0 kgf/cm²) suggests that higher HTHP treatment intensity promotes greater electron-donating and reducing activities in the ginseng extracts, likely due to the enhanced formation or release of redox-active compounds during thermal processing.
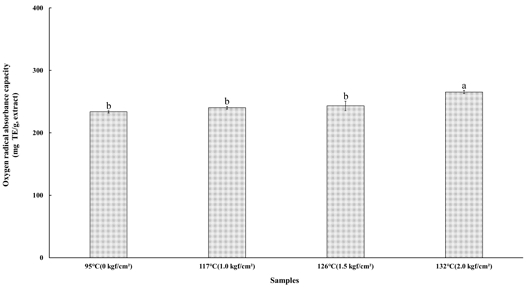
6. Oxygen Radical Absorbance Capacity Activity
The oxygen radical absorbance capacity (ORAC) assay is an inhibition-based method commonly employed to evaluate the total antioxidant capacity of a sample by quantifying the extent of oxidative damage caused by free radicals to a fluorescent probe (Jang HY et al 2008). In this assay, antioxidants present in the extract inhibit the free radical-induced decay of fluorescence in proportion to their concentration, thereby providing a reliable measure of the sample’s overall antioxidant potential. The ORAC activity of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure steaming treatments at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) is presented in Fig. 4. The ORAC values were 233.68±1.87 mg TE/g at 95℃ (0 kgf/cm²), 240.02±2.33 mg TE/g at 117℃ (1.0 kgf/cm²), 243.12±7.60 mg TE/g at 126℃ (1.5 kgf/cm²), and 265.43±2.44 mg TE/g at 132℃ (2.0 kgf/cm²). A significant increase in ORAC activity was observed with increasing temperature and pressure, following the order 132℃ (2.0 kgf/cm²) > 126℃ (1.5 kgf/cm²) > 117℃ (1.0 kgf/cm²) > 95℃ (0 kgf/cm²). The highest ORAC value was obtained under the 132℃ (2.0 kgf/cm²) condition, indicating that elevated HTHP treatment intensity enhanced the peroxyl radical scavenging capacity of the steamed cultivated wild ginseng extracts. Similarly, Brudzynski K & Miotto D (2011) reported higher ORAC values in thermally processed honeys (buckwheat, manuka, and dandelion) compared to their non-heated counterparts, while Pastoriza S & Rufián-Henares JA (2014) demonstrated that melanoidins exhibit greater scavenging activity against hydroxyl radicals than peroxyl radicals. This trend aligns with the present findings, where the DPPH and ORAC assays showed consistent yet distinct antioxidant patterns. Specifically, the DPPH assay measures antioxidant capacity via electron transfer mechanisms (Thaipong K et al 2006), whereas the ORAC assay quantifies hydrogen atom transfer (HAT) processes based on fluorescein oxidation kinetics (Sakurai S et al 2022).
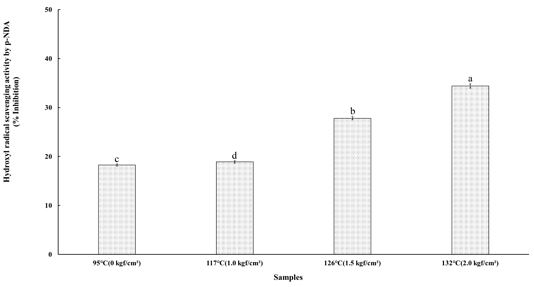
7. Hydroxyl Radical Scavenging Activity by p-NDA
The hydroxyl radical scavenging activity was evaluated based on the inhibition of p-nitrosodimethylaniline (p-NDA) bleaching. In this assay, hydroxyl radicals generated through the Fenton reaction selectively degrade p-NDA, and the scavenging capacity is determined by comparing the extent of bleaching in the presence and absence of the extract (Kunchandy E & Rao MNA 1990). The hydroxyl radical scavenging activity of the steamed cultivated wild ginseng extracts subjected to high-temperature and high-pressure steaming treatments at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²) is presented in Fig. 5. The scavenging activity values were 18.25% at 95℃ (0 kgf/cm²), 18.90% at 117℃ (1.0 kgf/cm²), 27.80% at 126℃ (1.5 kgf/cm²), and 34.40% at 132℃ (2.0 kgf/cm²). A statistically significant increase (p<0.05) in hydroxyl radical scavenging activity was observed with increasing temperature and pressure, following the order 132℃ (2.0 kgf/cm²) > 126℃ (1.5 kgf/cm²) > 117℃ (1.0 kgf/cm²) > 95℃ (0 kgf/cm²). The highest activity was recorded under the 132℃ (2.0 kgf/cm²) condition, suggesting that more intense HTHP treatments enhanced the hydroxyl radical scavenging efficiency of the steamed cultivated wild ginseng extracts.
CONCLUSION
This study investigated the effects of high-temperature and high-pressure (HTHP) steaming treatments on the antioxidant properties of cultivated wild ginseng. The steaming process was conducted at 95℃ (0 kgf/cm²), 117℃ (1.0 kgf/cm²), 126℃ (1.5 kgf/cm²), and 132℃ (2.0 kgf/cm²). The resulting extracts were analyzed for their total phenolic and flavonoid contents, as well as their antioxidant capacities, using DPPH, ABTS, FRAP, ORAC, and hydroxyl radical scavenging assays. The total phenolic and flavonoid contents increased progressively with rising temperature and pressure, reaching maximum levels of 6.04 mg GAE/g and 2.11 mg QE/g, respectively, under the 132℃ (2.0 kgf/cm²) condition. Similarly, the antioxidant activities—DPPH (14.42 mM TE/g), ABTS (68.94 mM TE/g), FRAP (675.75 μM TE/g), ORAC (265.43 mg TE/g), and hydroxyl radical scavenging activity (34.40%)—showed significant enhancements (p<0.05) with increasing processing intensity. These improvements are likely attributed to the thermal degradation of complex phenolic compounds into simpler, more active forms; the formation of low-molecular-weight antioxidants; and the production of Maillard reaction products, such as melanoidins, which exhibit potent reducing and radical scavenging abilities. In conclusion, HTHP steaming substantially enhanced the antioxidant potential of cultivated wild ginseng by increasing its phenolic and flavonoid contents and by strengthening multiple antioxidant mechanisms involving both electron transfer and hydrogen atom transfer pathways. These findings suggest that processing at 132℃ (2.0 kgf/cm²) optimizes the antioxidant efficacy of cultivated wild ginseng, providing a scientific foundation for the development of functional ginseng products with improved bioactivity and potential health-promoting effects.
Acknowledgments
This work was supported by the Kyungnam University Foundation Grant, 2025.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
References
-
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal Biochem 239(1): 70-76.
[https://doi.org/10.1006/abio.1996.0292]

-
Benzie IFF, Szeto YT (1999) Total antioxidant capacity of teas by the ferric reducing/antioxidant power assay. J Agric Food Chem 47(2): 633-636.
[https://doi.org/10.1021/jf9807768]

-
Brudzynski K, Miotto D (2011) The recognition of high molecular weight melanoidins as the main components responsible for radical-scavenging capacity of unheated and heat-treated Canadian honeys. Food Chem 125(2): 570-575.
[https://doi.org/10.1016/j.foodchem.2010.09.049]

-
Choi Y, Lee SM, Chun J, Lee HB, Lee J (2006) Influence of heat treatment on the antioxidant activities and polyphenolic compounds of Shiitake (Lentinus edodes) mushroom. Food Chem 99(2): 381-387.
[https://doi.org/10.1016/j.foodchem.2005.08.004]

-
Delgado-Andrade C, Rufián-Henares JA, Morales FJ (2005) Assessing the antioxidant activity of melanoidins from coffee brews by different antioxidant methods. J Agric Food Chem 53(20): 7832-7836.
[https://doi.org/10.1021/jf0512353]

-
Dewanto V, Wu X, Liu RH (2002) Processed sweet corn has higher antioxidant activity. J Agric Food Chem 50(17): 4959-4964.
[https://doi.org/10.1021/jf0255937]

-
Gillis CN (1997) Panax ginseng pharmacology: A nitric oxide link?. Biochem Pharmacol 54(1): 1-8.
[https://doi.org/10.1016/S0006-2952(97)00193-7]

-
Hong HD, Kim YC, Rho JH, Kim KT, Lee YC (2007) Changes on physicochemical properties of Panax ginseng C. A. Meyer during repeated steaming process. J Ginseng Res 31(4): 222-229.
[https://doi.org/10.5142/JGR.2007.31.4.222]

-
Huang D, Ou B, Hampsch-Woodill M, Flanagan JA, Prior RL (2002) High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J Agric Food Chem 50(16): 4437-4444.
[https://doi.org/10.1021/jf0201529]

-
Huang D, Ou B, Prior RL (2005) The chemistry behind antioxidant capacity assays. J Agric Food Chem 53(6): 1841-1856.
[https://doi.org/10.1021/jf030723c]

- Hwang IG, Woo KS, Kim TM, Kim DJ, Yang MH, Jeong HS (2006) Change of physicochemical characteristics of Korean pear (Pyrus pyrifolia Nakai) juice with heat treatment conditions. Korean J Food Sci Technol 38(3): 342-347.
-
Ichikawa K, Sasada R, Chiba K, Gotoh H (2019) Effect of side chain functional groups on the DPPH radical scavenging activity of bisabolane-type phenols. Antioxidants 8(3): 65.
[https://doi.org/10.3390/antiox8030065]

-
Jang HY, Park HS, Kwon KR, Rhim TJ (2008) A study on the comparison of antioxidant effects among wild ginseng, cultivated wild ginseng, and cultivated ginseng extracts. J Pharmacopunct 11(3): 67-78.
[https://doi.org/10.3831/KPI.2008.11.3.067]

-
Jeong SM, Kim SY, Kim DR, Jo SC, Nam KC, Ahn DU, Lee SC (2004) Effect of heat treatment on the antioxidant activity of extracts from citrus peels. J Agric Food Chem 52(11): 3389-3393.
[https://doi.org/10.1021/jf049899k]

- Jo HK, Sung MC, Ko SK (2011) The comparison of ginseng prosapogenin composition and contents in red and black ginseng. Kor J Pharmacogn 42(4): 361-365.
-
Kim JH, Nah SY (2005) Effects of ginsenoside Rg3 epimers on swine coronary artery contractions. J Ginseng Res 29(3): 119-125.
[https://doi.org/10.5142/JGR.2005.29.3.119]

-
Kim JS, Lee YS (2009) Antioxidant activity of melanoidins from different sugar/amino acid model systems: Influence of the enantiomer type. Food Sci Technol Int 15(3): 291-297.
[https://doi.org/10.1177/1082013209342916]

-
Kim WY, Kim JM, Han SB, Lee SK, Kim ND, Park MK, Kim CK, Park JH (2000) Steaming of ginseng at high temperature enhances biological activity. J Nat Prod 63(12): 1702-1704.
[https://doi.org/10.1021/np990152b]

-
Ko HM, Eom TK, Kim KC, Kim CJ, Lee JG, Kim JS (2018) Antioxidant effects and tyrosinase and elastase inhibitory activities of mountain ginseng adventitious roots extracts at different ethanol concentrations. Korean Journal of Agricultural Science 45(3): 499-508.
[https://doi.org/10.7744/kjoas.20180034]

-
Kunchandy E, Rao MNA (1990) Oxygen radical scavenging activity of curcumin. Int J Pharm 58(3): 237-240.
[https://doi.org/10.1016/0378-5173(90)90201-E]

-
Kwak YS, Choi KH, Kyung JS, Won JY, Rhee MH, Lee JG, Hwang MS, Kim SC, Park CK, Song KB, Han GH (2008) Effect of high temperature heating on the some physicochemical properties of Korean red ginseng (Panax ginseng C. A. Meyer) water extract. J Ginseng Res 32(2): 120-126.
[https://doi.org/10.5142/JGR.2008.32.2.120]

- Kwon OC, Woo KS, Kim TM, Kim DJ, Hong JT, Jeong HS (2006) Physicochemical characteristics of garlic (Allium sativum L.) on the high temperature and pressure treatment. Korean J Food Sci Technol 38(3): 331-336.
- Lee JH, Shen GN, Kim EK, Shin HJ, Myung CS, Oh HJ, Kim DH, Roh SS, Cho W, Seo YB, Park YJ, Kang CW, Song GY (2006) Preparation of black ginseng and its antitumor activity. J Physiol & Pathol Korean Med 20(4): 951-956.
-
Lim W, Mudge KW, Vermeylen F (2005) Effects of population, age, and cultivation methods on ginsenoside content of wild American ginseng (Panax quinquefolium). J Agric Food Chem 53(22): 8498-8505.
[https://doi.org/10.1021/jf051070y]

-
Nam KY, Lee NR, Moon BD, Song GY, Shin HS, Choi JE (2012) Changes of ginsenosides and color from black ginsengs prepared by steaming-drying cycles. Korean J Medicinal Crop Sci 20(1): 27-35.
[https://doi.org/10.7783/KJMCS.2012.20.1.027]

- Park JH (2004) Sun ginseng-A new processed ginseng with fortified activity. Food Ind Nutr 9(1): 23-27.
-
Pastoriza S, Rufián-Henares JA (2014) Contribution of melanoidins to the antioxidant capacity of the Spanish diet. Food Chem 164: 438-445.
[https://doi.org/10.1016/j.foodchem.2014.04.118]

-
Patrignani M, González-Forte LDS (2021) Characterisation of melanoidins derived from Brewers’ spent grain: new insights into their structure and antioxidant activity. Int J Food Sci Technol 56(1): 384-391.
[https://doi.org/10.1111/ijfs.14653]

-
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26(9-10): 1231-1237.
[https://doi.org/10.1016/S0891-5849(98)00315-3]

-
Sakurai S, Kikuchi A, Gotoh H (2022) Hydrophilic oxygen radical absorbance capacity values of low-molecular-weight phenolic compounds containing carbon, hydrogen, and oxygen. RSC Adv 12(7): 4094-4100.
[https://doi.org/10.1039/D1RA08918H]

-
Saura-Calixto F (2011) Dietary fiber as a carrier of dietary antioxidants: an essential physiological function. J Agric Food Chem 59(1): 43-49.
[https://doi.org/10.1021/jf1036596]

-
Hu SY (1976) The genus Panax (Ginseng) in Chinese medicine. Econ Bot 30(1): 11-28.
[https://doi.org/10.1007/BF02866780]

-
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3): 144-158.
[https://doi.org/10.5344/ajev.1965.16.3.144]

-
Srinivasan R, Chandrasekar MJN, Nanjan MJ, Suresh B (2007) Antioxidant activity of Caesalpinia digyna root. J Ethnopharmacol 113(2): 284-291.
[https://doi.org/10.1016/j.jep.2007.06.006]

-
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Byrne DH (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compos Anal 19(6-7): 669-675.
[https://doi.org/10.1016/j.jfca.2006.01.003]

-
Tores de la Cruz S, Iriondo-DeHond A, Herrera T, Lopez-Tofiño Y, Galvez-Robleño C, Prodanov M, Velazquez-Escobar F, Abalo R, Castillo MDD (2019) An assessment of the bioactivity of coffee silverskin melanoidins. Foods 8(2): 68.
[https://doi.org/10.3390/foods8020068]

-
Woisky RG, Salatino A (1998) Analysis of propolis: some parameters and procedures for chemical quality control. J Apic Res 37(2): 99-105.
[https://doi.org/10.1080/00218839.1998.11100961]

- Woo KS, Jang KI, Kim KY, Lee HB, Jeong HS (2006) Antioxidative activity of heat treated licorice (Glycyrrhiza uralensis Fisch) extracts. Korean J Food Sci Technol 38(3): 355-360.
- Yang SJ, Woo KS, Yoo JS, Kang TS, Noh YH, Lee JS, Jeong HS (2006) Change of Korean ginseng components with high temperature and pressure treatment. Korean J Food Sci Technol 38(4): 521-525.
-
Yun TK (2001) Panax ginseng-a non-organ-specific cancer preventive? Lancet Oncol 2(1): 49-55.
[https://doi.org/10.1016/S1470-2045(00)00196-0]